Patented MDI Technology
Respitab is an innovative tablet-based technology platform designed for use in pressurized metered dose inhalers (pMDIs). It offers a novel approach to inhalation product formulation and manufacturing. Unlike conventional pMDI suspension processes—which require complex mixing and homogenization of drug particles in propellant—Respitab enables the drug and excipients to be compressed into a solid tablet (the “Respitab”), which is then placed directly into the inhaler canister.
Key Features and Advantages

Key Features and Advantages
The Respitab process eliminates the need for high-shear mixing, homogenization, and continuous agitation during filling. Tablets are produced using standard tablet presses and are simply dropped into canisters before propellant filling—streamlining production and reducing equipment needs.

Enhanced Batch Uniformity
Each Respitab tablet contains a pre-measured dose of active pharmaceutical ingredient (API) and excipients. This ensures batch uniformity and minimizes batch-to-batch variability.

Reduced API Loss and Waste
By eliminating bulk suspension handling and cleaning steps, Respitab minimizes API loss and cross-contamination risk—making it ideal for costly or potent drugs.

Flexible and Scalable
The technology supports flexible batch sizes and is easily scalable. It enables rapid adaptation to different production volumes and facilitates straightforward tech transfer between manufacturing sites.

Improved Stability
Since the API is stored in solid state until the propellant is added, the risk of sedimentation or aggregation is significantly reduced compared to traditional pMDI suspensions.
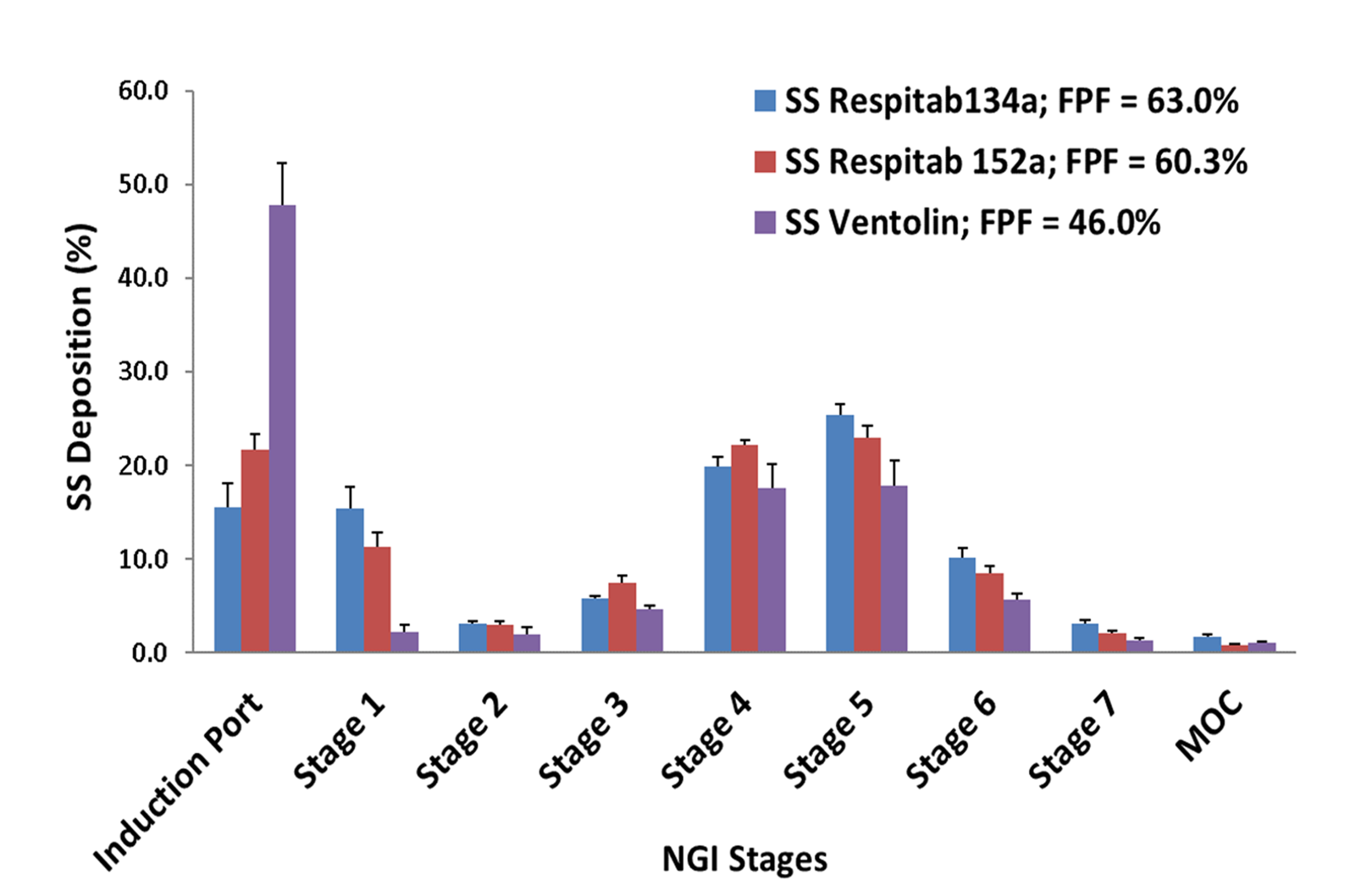
Respitab™ – Example Of Salbutamol PMDI Performance